Lipids
Lipids are a family of compounds whose diversity is also made possible by building complex molecules from multiple units of simpler molecules, and once again one sees characteristic rings and chains.
Polar and Non-polar Molecules
You have probably heard the expression "oil and water don't mix," and you have observed how salad dressing composed of vinegar (which is aqueous, i.e., largely water) and oil will separate when left to stand. This incompatibility is due to the fact that water molecules are polar, but oil is non-polar. Water is a polar molecule because the negatively charged electrons that spin around the nuclei of the atoms are not evenly distributed. The oxygen atom has much more mass than the two hydrogen atoms, and therefore the electrons spend more time in the vicinity of the oxygen atom. As a result, the end of the water molecule where oxygen is located is relatively negative in charge, whereas the end with hydrogens is relatively positively charged. The positive ends of the water molecule are attracted to the negative ends of adjacent water molecules, as shown in the figure below, and this enables water molecules to coalesce. You may have also seen water bead on a car windshield as a result of this phenomenon.
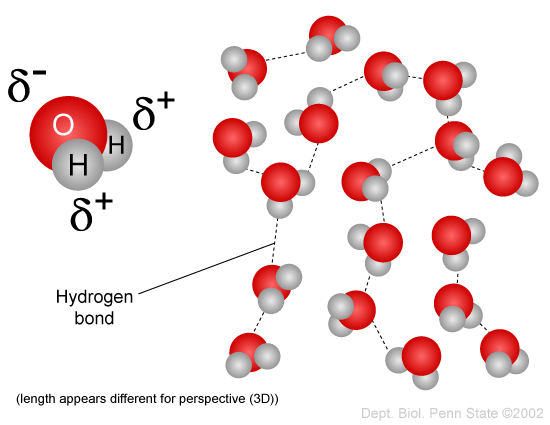
Source: http://www.personal.psu.edu/staff/m/b/mbt102/bisci4online/chemistry/chemistry3.htm
Lipids, i.e., fatty molecules, on the other hand, are non-polar, meaning that the charge distribution is evenly distributed, and the molecules do not have positive and negatively charged ends.. Non-polar molecules do not dissolve well in polar solutions like water; in fact, polar and non-polar molecules tend to repel each other in the same way that oil and water don't mix and will separate from each other even if they are shaken vigorously in an attempt to mix them. This distinction between polar and non-polar molecules has important consequences for living things, which are composed of both polar molecules and non-polar molecules. The next sections will illustrate the importance of this.
Fatty Acids
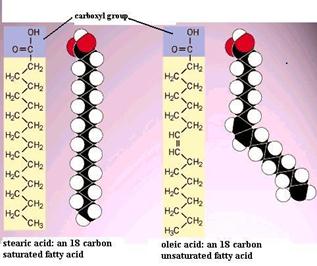
Fatty acids are chain-like molecules that are important components of several types of lipids. The illustrations below show two different fatty acid molecules. Each has a characteristic carboxyl group (the -COOH) attached to a chain of carbons with hydrogen atoms attached to the carbon chain. Two things are noteworthy. First, the hydrocarbon chain is very non-polar and therefore doesn't dissolve in water very well. However, hydrocarbon chains do associate with each other readily. Second, note that the unsaturated fatty acid has two hydrogens removed, and this allows formation of a double bond, i.e., a stronger bond between two of the carbon atoms. Note also that the double bond tends to produce a bend or a kink in the fatty acid. The illustration to the right shows two other common fatty acids: stearic acid, which is a straight 18 carbon chain with no double bonds, and oleic acid, which is an 18 carbon chain with a single double bond, which cause a bend in the carbon chain.
Triglycerides
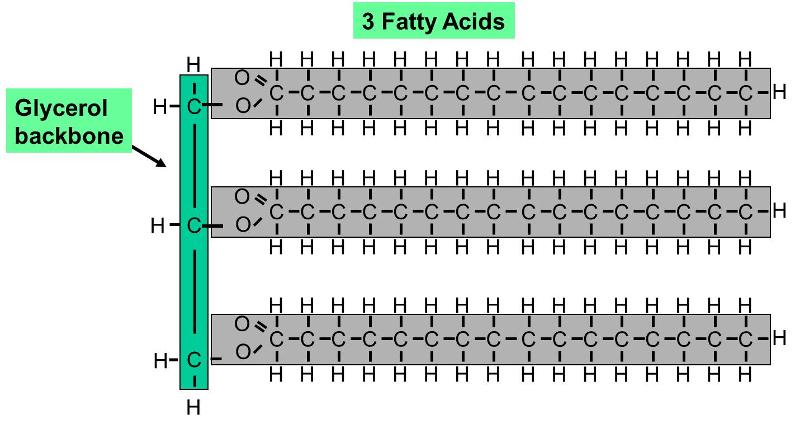
A fat molecule is a type of lipid that consists of three fatty acid molecules connected to a 3 carbon glycerol backbone, as shown on the right. The three fatty acids can be different from one another. Since the hydrocarbon chains are very non-polar, fats do no dissolve in water; instead, fat molecules tend to coalesce with one another. Since a fat molecule has 3 fatty acids connected to a glycerol molecule, they are also called trigylcerides.
Phospholipids
Phospholipids constitute another important class of lipids. These are similar to similar to trigylcerides in that they have a glycerol backbone, but there are only two fatty acids connected to glycerol. The third carbon of the glycerol backbone is attached to a phosphate group (an atom of phosphorus bonded to four atoms of oxygen), and the phosphate group is attached to a base molecule of choline, serine, or ethanolamine. The part of the phospholipid with phosphate and the base is actually very polar, and it tends to rotate away from the two fatty acids. This makes phospholipid molecules have a hairpin shape. The head of the hairpin is very polar and therefore likes to associate with water (it is hydrophilic), while the two fatty acid chains (the "tails") are very non-polar and tend to avoid water (hydrophobic) and associate with other hydrocarbon chains.
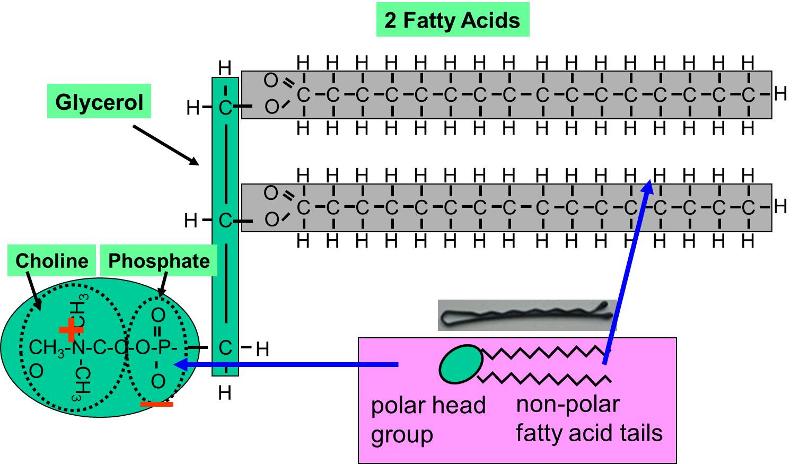
Phospholipids can be described as amphipathic ("amphi" means "both"), because they have this dual nature (part polar and part non-polar). This characteristic causes phospholipids to self-associate into large macromolecular complexes in an aqueous (watery) environment.
Cholesterol
Cholesterol is also an important component of animal membranes (plant membranes have a similar, but distinct 'sterol' in their membranes). It is a lipid, because it is composed almost entirely of carbon and hydrogen, but it is different from fatty acids, fats and phospholipids in that it is arranged in a series of rings. The rings consist of 5 or 6 carbon atoms bonded together. The carbon atoms at the apices of the hexagonal and pentagonal rings have hydrogen atoms attached to them. The ring-like structures are fairly rigid, but there is also a hydrocarbon tail, which is somewhat flexible. The entire structure is somewhat reminiscent of a fancy kite with a tail.
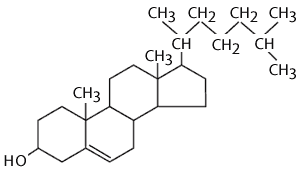
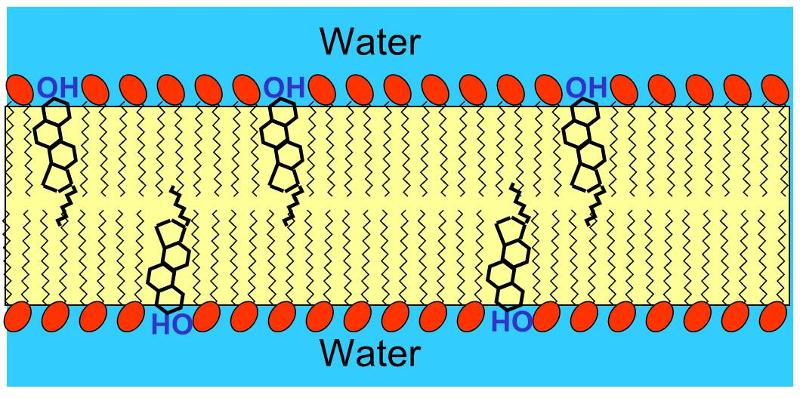
Cholesterol is very non-polar, except for the hydroxyl group attached to the first ring. Consequently, in an animal cell membrane the polar hydroxyl group sticks into the aqueous environment (either extracellular water or intracellular water), and the rest of the cholesterol molecule, which is non-polar, is found among the non-polar fatty acid tails of the phospholipids.The image below depicts a section of a cell membrane with water outside and inside. The polar head groups of the phospholipids are represented in red, and their non-polar fatty acid tails are shown as zig-zag lines extending from the polar head group. As we we see in greater detail, cell membranes consist of a bilayer of phospholipids with other molecules inserted into the bilayer. This illustration shows five cholesterol molecules (the black structures with four conjoined rings) inserted into the lipid bilayer. Most of the cholesterol molecule in non-polar and therefore associations with the non-polar fatty acid tails of the phospholipids. However, the hydroxyl group (-OH) on cholesterol carries a negative charge and therefore associates with the polar environment of water either inside the cell or outside.