More Complex Biological Molecules
Carbohydrates
Sugar Molecules
The stuff of life is amazingly diverse and complex, but it is all based on combinations of simple biological molecules. Biological molecules are often made from chains & rings of carbon. These molecular structures can be represented by "stick drawings" that show the component atoms (e.g., C, H, N, O for carbon, hydrogen, nitrogen, and oxygen respectively) and show the bonds between them as dashes. A single dash ( - ) represents a single bond, and a double dash (=) represents a double bond.
Note that some common "groups" are depicted without showing the bonds between them. For example, the hydroxyl group (-OH) consists of a hydrogen atom bonded to an oxygen atom:
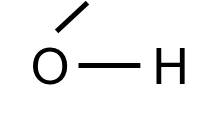
The hydoxyl group will commonly be bonded to a carbon atom in this fashion:
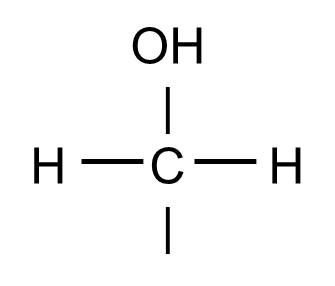
And this structure might be found, for example, as part of a glucose molecule, depicted below.
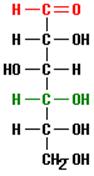
This molecule of the sugar glucose consists of 6 carbon atoms bonded together as a chain with additional atoms of oxygen and hydrogen. Note that the previous structure (a carbon to which two hydrogens and one hydroxyl group are bound) is located at the bottom of this glucose chain where it is written using the notation CH2OH.
consists of 6 carbon atoms bonded together as a chain with additional atoms of oxygen and hydrogen. Note that the previous structure (a carbon to which two hydrogens and one hydroxyl group are bound) is located at the bottom of this glucose chain where it is written using the notation CH2OH.
This glucose chain forms a ring in aqueous solutions, e.g., in body fluids, as shown below.
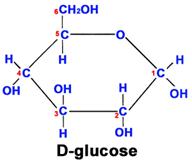
Fructose is another sugar that also has 6 carbons, 12 hydrogens, and 6 oxygen atoms. However, the arrangement of the atoms is different, and this makes it much sweeter than glucose and also affects its ability to combine with other molecules.
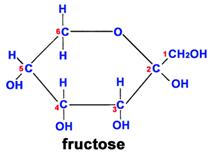
Another important theme is that single units of biological molecules (monomers) can join to form increasingly complex molecules (polymers). For example, two monosaccharide sugars can also become bound together chemically to form a disaccharide. Sucrose is the disaccharide in common sugar that we buy at the grocery store. The structure of sucrose is shown below.
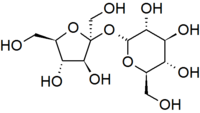
Polysaccharides: Starch, Glycogen, and Cellulose
Glucose and fructose are examples of monosaccharides, meaning they consist of a single sugar unit, while sucrose is an example of a disaccharide. However, sugar units can be bonded or linked together to form polysaccharides , which consist of many sugars linked together to form extensive chains of sugars. Plants store energy as starch
, which consist of many sugars linked together to form extensive chains of sugars. Plants store energy as starch , which consists of very long chains of glucose linked together.
, which consists of very long chains of glucose linked together.
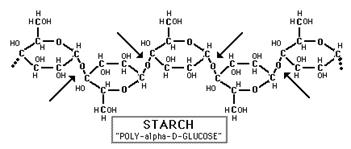
Source: http://www.science-projects.com/Amylase.htm
Animals store energy as glycogen , which consists of more highly branched chains of glucose. Collectively, sugars, starch, and glycogen are know as carbohydrates, and they are an important source of cellular energy.
, which consists of more highly branched chains of glucose. Collectively, sugars, starch, and glycogen are know as carbohydrates, and they are an important source of cellular energy.
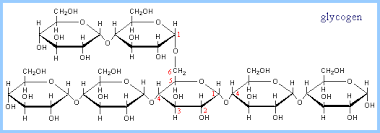
Source: https://www.rpi.edu/dept/bcbp/molbiochem/MBWeb/mb1/part2/glycogen.htm
Cellulose i s yet another polysaccharide formed from glucose. Cellulose is composed of unbranched, parallel chains of glucose. A key feature is that the chains bond to one another to form strong fibers that serve a structural purpose. Humans do not have the enzymes necessary to break the bonds in cellulose, and any cellulose we ingest passes through our digestive systems. It is a major component of what we refer to as dietary "fiber."
s yet another polysaccharide formed from glucose. Cellulose is composed of unbranched, parallel chains of glucose. A key feature is that the chains bond to one another to form strong fibers that serve a structural purpose. Humans do not have the enzymes necessary to break the bonds in cellulose, and any cellulose we ingest passes through our digestive systems. It is a major component of what we refer to as dietary "fiber."
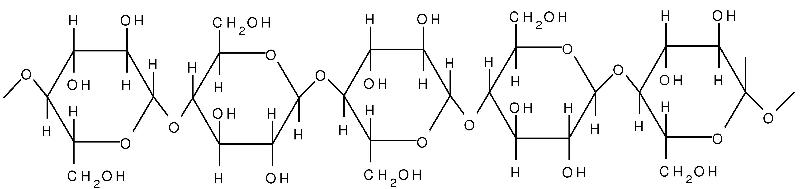
Source: http://www.intechopen.com/books/cellulose-fundamental-aspects/cellulose-microfibril-angle-in-wood-and-its-dynamic-mechanical-significance