Pathogenesis of Atherosclerosis
The Response to Injury Theory
The endothelial cells that line blood vessels provide an active, dynamic interface between the blood stream and the arterial wall. Their most obvious function is to provide a semi-permeable barrier that regulates the exchange of fluid, nutrients, gases, and waste between the blood and tissues. In addition, endothelial cells regulate a number of other less obvious processes. They provide a unique surface that generally allows the cellular elements of blood to flow without adhering to the vessel lining unless some insult has perturbed the cells. When perturbation occurs, endothelial cells secrete cytokines that trigger and maintain an inflammatory response. Endothelial cells also regulate constriction and relaxation of vessels by releasing vasodilatory molecules (e.g., nitric oxide (NO) and prostacyclin (PGI2) and vasoconstrictive molecules (endothelin and angiotensin-II). [For an excellent review, see Russell Ross: Cell biology of atherosclerosis. Annu. Rev. Physiol. 1995. 57791-804).
Atherosclerosis is a disease process which is triggered by sometimes subtle physical or chemical insults to the endothelial cell layer of arteries. The "Response to Injury Theory" now has widespread acceptance among scientific and medical scholars. This theory holds that the earliest event in atherogenesis is injury to the endothelium
is injury to the endothelium , which can be triggered by any number of insults, either alone or in combination. These include:
, which can be triggered by any number of insults, either alone or in combination. These include:
- Physical injury or stress as a result of direct trauma or hypertension

- Turbulent blood flow, for example, where arteries branch
- Circulation of reactive oxygen species (free radicals), e.g., from smoking or air pollutants
- Hyperlipidemia (high blood concentrations of LDL or VLDL)
- Chronically elevated blood glucose levels
- Homocysteinemia, which results from an inherited metabolic defect that leads to very high levels of the homocysteine, a metabolite of methionine; high concentrations are toxic to the endothelium.
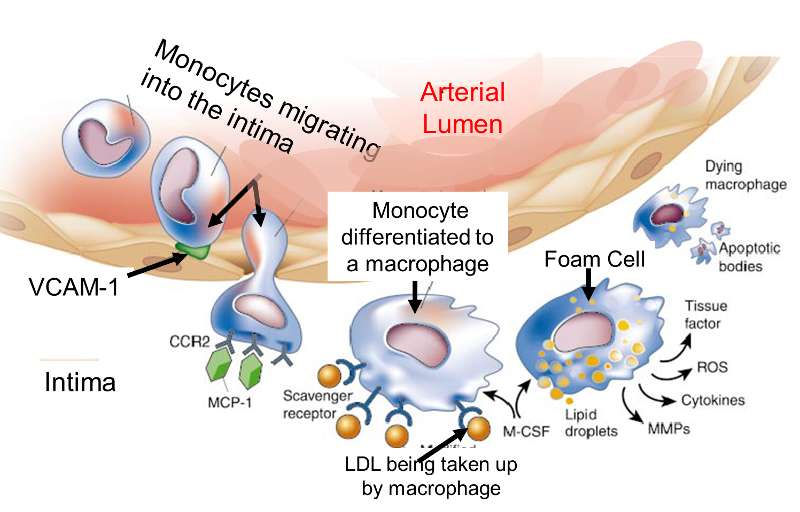
Under normal conditions, leukocytes in the blood do not adhere to the endothelial cells that line all blood vessels. However, injury to endothelial cells provokes an inflammatory response as illustratedbelow.
in the blood do not adhere to the endothelial cells that line all blood vessels. However, injury to endothelial cells provokes an inflammatory response as illustratedbelow.
Adapted from http://www.nature.com/nature/journal/v420/n6917/fig_tab/nature01323_F1.html
The endothelial cells begin to produce cell surface adhesion molecules such as VCAM-1, causing monocytes and T-lymphocytes to adhere to the endothelium and then migrate beneath it by squeezing between the endothelial cells. Circulating monocytes and T-lymphocytes are attracted to the sites of injury by chemoattractant cytokines (chemokines).
causing monocytes and T-lymphocytes to adhere to the endothelium and then migrate beneath it by squeezing between the endothelial cells. Circulating monocytes and T-lymphocytes are attracted to the sites of injury by chemoattractant cytokines (chemokines).
The endothelial cells also change shape, and the tight junctions between endothelial cells loosen, increasing the permeability to fluid, lipids, and leukocytes. Lipoprotein particles, and especially low-density lipoprotein (LDL), enter the arterial wall and undergo oxidation. Oxidation of LDL in the arterial wall occurs as a result of its exposure to nitric oxide, macrophages, and some enzymes such as lipoxygenase. Once they have migrated into the intima, monocytes differentiate into macrophages and begin to take up oxidized LDL that has gotten into the intima. Macrophages retain the lipid they take up, and as they become more lipid-laden, they are referred to as "foam cells." Eventually, the foam cell will undergo apoptosis and die, but the lipid will accumulate in the intima.
in the arterial wall occurs as a result of its exposure to nitric oxide, macrophages, and some enzymes such as lipoxygenase. Once they have migrated into the intima, monocytes differentiate into macrophages and begin to take up oxidized LDL that has gotten into the intima. Macrophages retain the lipid they take up, and as they become more lipid-laden, they are referred to as "foam cells." Eventually, the foam cell will undergo apoptosis and die, but the lipid will accumulate in the intima.
The Fatty Streak
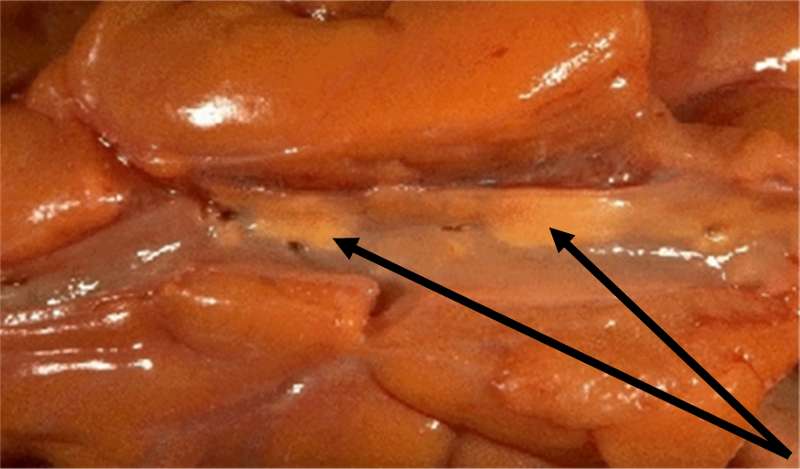
Fatty streaks are the first signs of atherosclerosis that are visible without magnification. They consist of lipid-containing foam cells in the arterial wall just beneath the endothelium. The image below on the left is a photograph of the inside of an artery; one can see two yellowish fatty streaks beneath the thin endothelial lining of the artery. These lesions occur in the aorta and coronary arteries of most people by age 20. Over time, they can evolve into atherosclerotic plaques or they can remain stable or even regress.
are the first signs of atherosclerosis that are visible without magnification. They consist of lipid-containing foam cells in the arterial wall just beneath the endothelium. The image below on the left is a photograph of the inside of an artery; one can see two yellowish fatty streaks beneath the thin endothelial lining of the artery. These lesions occur in the aorta and coronary arteries of most people by age 20. Over time, they can evolve into atherosclerotic plaques or they can remain stable or even regress.
Adapted from http://wwwf.imperial.ac.uk/~ajm8/BioFluids/Pictures/
The cartoon below summarizes the steps by which a fatty streak evolves. Injury to the endothelium triggers monocyte adhesion, a loosening of endothelial cell junctions, and migration of monocytes beneath the endolthelium where they differentiate into macrophages. The more permeable endothelium also permits LDL to enter the intima of the artery, and macrophages begin engulfing the LDL by phagocytosis. After macrophages become laden with lipid from ingesting LDL, they are referred to as "foam cells," and collections of these create fatty streaks.
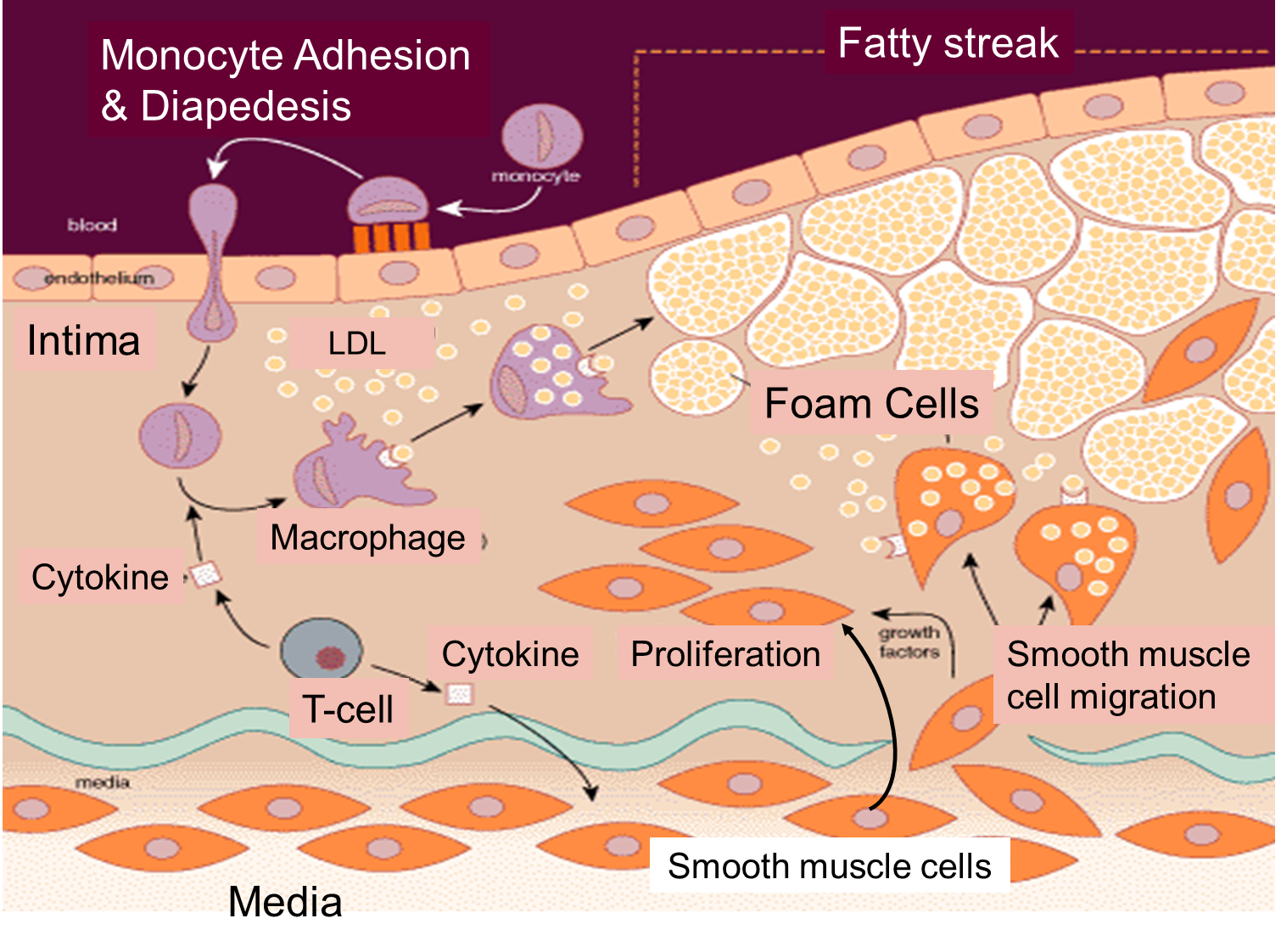
The cartoon also emphasizes the role of T-lymphocytes, which are also in the intima. They secrete cytokines that eventually induce smooth muscle cells to migrate from the media to the intima. These smooth muscle cells also begin to proliferate under the influence of growth factors. Over time there is a progressive accumulation of lipid and smooth muscle cells in the intima, and eventually the growing lesion begins to raise the endothelium and encroach on the lumen of the artery. This is depicted in the image below, which shows a cross-section of an artery at the site of an atherosclerotic plaque.
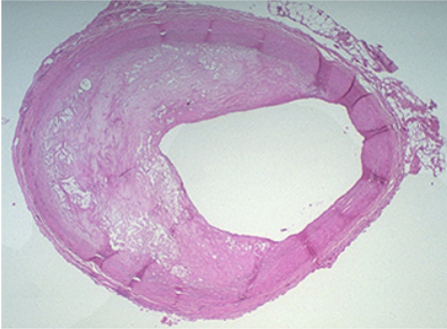
The right side of the artery has a fairly normal appearance, but an atherosclerotic plaque has evolved on the left side. The mass of the "atheroma" is composed of a mixture of lipid and subintimal smooth muscle cells. Note that much of the lumen of the artery is occupied by this growing lesion.
Stable & Unstable Plaques
The image below illustrates the evolution of atherosclerotic plaques and also indicates that there are two possible forms of evolution. Slowly growing plaques expand gradually due to accumulation of lipid in foam cells and migration and proliferation of smooth muscle cells. These plaques tend to stabilize and are not prone to rupture. The so-called fibrin cap on the lesion matures. In contrast, other plaques grow more rapidly as a result of more rapid lipid deposition. These have thin fibrin caps that are prone to rupture. Once a plaque ruptures, it can trigger an acute thrombosis (clot) by activating platelets and the clotting cascade.
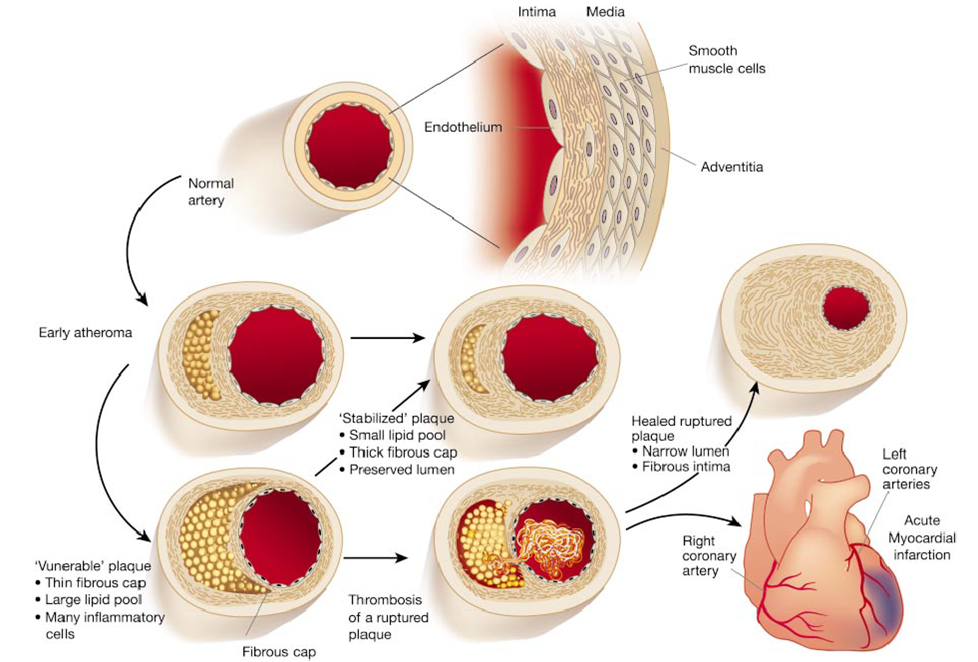
Illustration from from Libby P: Inflammation in Atherosclerosis. Nature 202;420:868