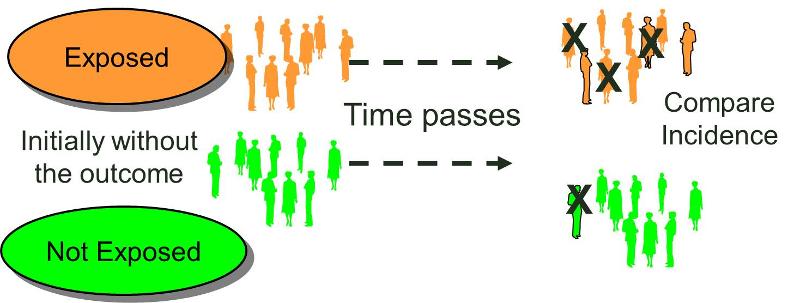
Cohort Studies

The characteristic feature of a cohort study is that the investigator identifies subjects at a point in time when they do not have the outcome of interest and compares the incidence of the outcome of interest among groups of exposed and unexposed (or less exposed) subjects. (We can refer to the groups being compared as exposure cohorts.) Cohorts may be identified retrospectively or prospectively, but in either case the outcome status needs to be established at least twice. It must be established that a cohort did not have the outcome of interest at the beginning of the observation period, and the cohort needs to be examined again to determine whether or not the outcome subsequently developed, i.e., the incidence in each of the exposure groups.
Upon successful completion of this section of the course, the student will be able to:
- Prospective cohort study
- Retrospective cohort study
- Ambidirectional study
- An internal comparison group
- An external comparison group
- A general population comparison group

There are two fundamental types of cohort studies based on when and how the subjects are enrolled into the study:
In prospective cohort studies the investigators conceive and design the study, recruit subjects, and collect baseline exposure data on all subjects, before any of the subjects have developed any of the outcomes of interest. The subjects are then followed into the future in order to record the development of any of the outcomes of interest. The follow up can be conducted by mail questionnaires, by phone interviews, via the Internet, or in person with interviews, physical examinations, and laboratory or imaging tests. Combinations of these methods can also be used.
Typically, the investigators have a primary focus, for example, to learn more about cardiovascular disease or cancer, but the data collected from the cohort over time can be used to answer many questions and test many possible determinants, even factors that they hadn't considered when the study was originally conceived.
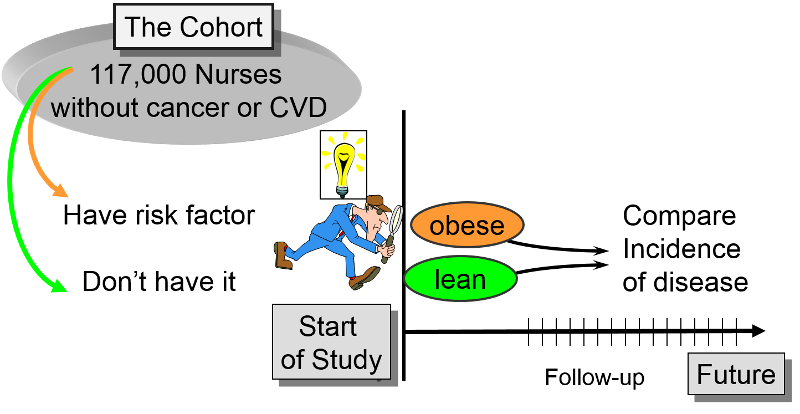
The Framingham Heart Study, the Nurses Health Study, and the Black Women's Health Study are good examples of large, productive prospective cohort studies. In each of these studies, the investigators wanted to study risk factors for common chronic diseases. The investigators identified a cohort (group) of possible subjects who would be feasible to follow for a prolonged period. Eligible subjects had to meet certain criteria (inclusion criteria) to be included in the study as subjects. The investigators then determine the initial or "baseline" characteristics, behaviors, and other "exposures" of all subjects at the beginning of the study. Information is collected from all subjects in the same way using exactly the same questions and data collection methods for all subjects. They design the questions and data collection procedures very carefully in order to have accurate information about exposures before disease develops in any of the subjects.
For more information:
Link to Framingham Heart Study
Link to The Nurses Health Study
Link to The Black Women's Health Study
Of course, data analysis cannot take place until enough 'events' or 'outcomes' have occurred, so time must elapse, and the analyses will look at events that have occurred during the period of time from the beginning of the study until the time of the analysis or the end of the study. It goes without saying that analysis is always done retrospectively, because a span of time has to have elapsed before you can compare incidence. The thing that makes prospective cohort studies prospective is that they were designed prospectively, and subjects were enrolled and had baseline data collected before any of them developed any of the outcomes of interest. Determining baseline exposure status before disease events occur gives prospective studies an important advantage in reducing certain types of bias that can occur in retrospective cohort studies and case-control studies, though at the cost of efficiency.
After baseline information is collected, subjects in a prospective cohort study are then followed "longitudinally," i.e. over a period of time, usually for years. This enables the investigators to know when follow up began, if and when subjects become diseased, if and when they become lost to follow up, and whether their exposure status changed during the follow up period. By having individual data on these details for each subject, the investigators can compute and compare the incidence rates for each of the exposure groups.
The illustration below shows a hypothetical group of 12 subjects followed over a number of years. They were enrolled into the study at different times, and some of them became lost to follow up, i.e., they stopped responding to letters, emails and phone calls, so we don't know what happened to them; these are show by the horizontal follow up line stopping.

Three subjects developed the outcome of interest at the approximate dates show by the "X"s. The incidence rate was calculated by computing the disease free observation time for each subject, adding up the disease-free observation times for the entire group, and then dividing this into the number of events, as shown in the calculation below the time line.
Since the investigators asked about many exposures during baseline data collection, they can eventually use the data to study many associations between different exposures and disease outcomes. For example, one could identify smokers and non-smokers at baseline and compare their subsequent incidence of developing heart disease. Alternatively, one could group subjects based on their body mass index (BMI) and compare their risk of developing heart disease or cancer.
The data in the table below summarizes some of the results in a study is from the Nurses' Health Study in which they examined the association of body mass index (BMI) with heart disease. (Link to the article)
|
Body Mass Index |
# Non-fatal Heart Attacks |
Person-Years of Observation |
MI Rate per 100,000 Person-Years |
Rate Ratio |
|
<21 |
41 |
177,356 |
23.1 |
1.0 |
|
21-23 |
57 |
194,243 |
29.3 |
1.3 |
|
23-25 |
56 |
155,717 |
36.0 |
1.6 |
|
25-29 |
67 |
148,541 |
45.1 |
2.0 |
|
>29 |
85 |
99,573 |
85.4 |
3.7 |
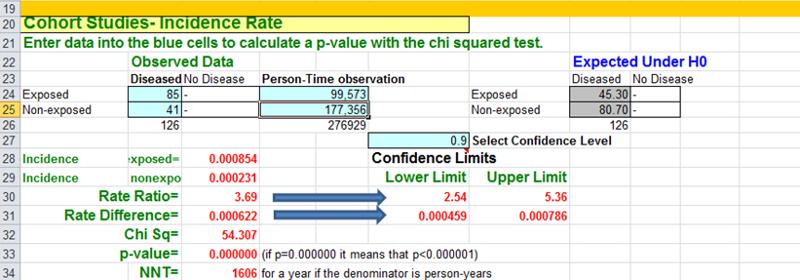
There were over 118,000 nurses in the study, and they divided the cohort int0 five exposure groups based on BMI. In this case they used the incidence rate of myocardial infarctions (MI, i.e., heart attacks) in the leanest women (BMI < 21) as a reference, against which they compared the incidence rates of MI in the other four groups. For example, the incidence rate of MI in the reference group (those with BMI < 21) was 23.1 per 100,000 person-years of disease-free observation time. The incidence rate in the heaviest group (BMI > 29) was 85.4 MIs per 100,000 person-years.
The Epi-Tools.XLS worksheet for cohort studies can compare either cumulative incidence (top section of the worksheet) or incidence rates like these (lower section of the worksheet). For example, if one were to compare the heaviest group (BMI > 29) to the women with BMI < 21 (the reference group), the Epi-Tools analysis would look like this
Manson et al. also used the Nurses' Health Study (NHS) to examine the effect of exercise on cardiovascular disease. The NHS enrolled 121,700 female RNs in 1976, but they didn't begin to collect information on exercise until 1986. Since the original baseline data did not include information on exercise, the exercise study only used the women who had not yet developed any cardiovascular problems by 1986. So, the exercise study was restricted to the 72,448 subjects who were free of cardiovascular disease and cancer in 1986. In essence, the information on exercise and activity that they began collecting in 1986 represented a new baseline for this subset of the original cohort.
Link to the article by Manson et al.
Retrospective studies also group subjects based on their exposure status and compare their incidence of disease. However, in this case both exposure status and outcome are ascertained retrospectively.
In essence, the investigators jump back in time to identify a useful cohort which was initially free of disease and 'at risk.' They then use whatever records are available to determine each subject's exposure status at the begin of the observation period, and they then ascertain what subsequently happened to the subjects in the two (or more) exposure groups. Retrospective cohort studies are also 'longitudinal,' because they examine health outcomes over a span of time. The distinction is that in retrospective cohort studies all of the cases of disease have already occurred before the investigators initiate the study. In contrast, exposure information is collected at the beginning of prospective cohort studies before any subjects have developed any of the outcomes or interest, and the 'at risk' period begins after baseline exposure data is collected and extends into the future.
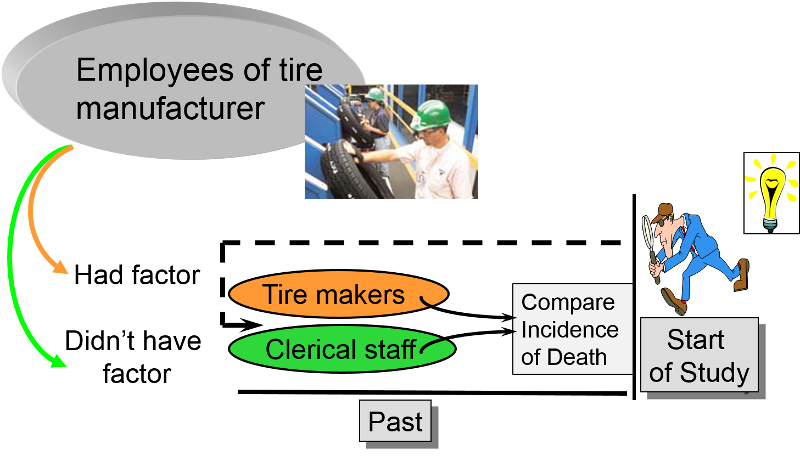
Retrospective cohort studies are particularly useful for unusual exposures or occupational exposures. For example, if an investigator wanted to determine whether exposure to chemicals used in tire manufacturing was associated with an increased risk of death, one might find a tire manufacturing factory that had been in operation for several decades. One could potentially use employee health records to identify those who had had jobs which involved exposure to the chemicals in question (e.g., workers who actually manufactured tires) and non-exposed coworkers (e.g., clerical workers or sales personnel in the same company or, even better, workers also involved in manufacturing operations but with jobs that didn't involve exposure to the chemicals). One could then ascertain what had happened to all the subjects and compare the incidence of death in the exposed and non-exposed workers.
Retrospective cohort studies like this are very efficient because they take much less time and cost much less than prospective cohort studies, but this advantage also creates potential problems. Sometimes exposure status is not clear when it is necessary to go back in time and use whatever data was available, because the data being used was not designed to be used in a study. Even if it was clear who was exposed to tire manufacturing chemicals based on employee records, it would also be important to take into account (or adjust for) other differences that could have influenced mortality (confounding factors). For example, in a study comparing mortality rates between workers exposed to solvents used in tire manufacture and an unexposed comparison group, it might be important to adjust for confounding factors such as smoking and alcohol consumption. However, it is unlikely that a retrospective cohort study would have accurate information on these other risk factors.
When an outbreak of Giardia (see this Link to CDC page on Giardia) occurred in Milton, MA , the Milton Health Department requested assistance from the epidemiologists in the MA Department of Public Health. (Kathleen MacVarish from the BUSPH Practice Office was the Health Agent in Milton who led the investigation.) The request for assistance was made some time after the start of the outbreak, and the outbreak was winding down by the time DPH began their study. The outbreak was clearly concentrated among members of the Wollaston Golf Club in Milton, MA , which had two swimming pools, one for adults and a wading pool for infants and small children. Given what they knew about the usual mechanisms by which Giardia is transmitted, the investigators thought that contamination of the kiddy pool by a child shedding Giardia into their stool was the most likely source. (NOTE) The study was conducted by getting most of the people in the cohort to complete a questionnaire in which one of the key questions was "Did you spend any time in the kiddy pool?" This outbreak clearly took place in a well-defined cohort (members of the club), and the investigators could determine how many people developed Giardia in each of the exposure groups (i.e., exposed to the kiddy pool or not). Moreover, they also knew how many respondents had been exposed to the kiddy pool and how many were not. In other words, they knew the denominators for the exposure groups, so they could calculate the cumulative incidence, risk difference, and the risk ratio. They found that people who had spent time in the kiddy pool had 9.0 more cases per 100 persons than those who spent time in the kiddy pool. The risk ratio was 3.27. Because the investigation started after the cases had already occurred, DPH's study of Giardia in Milton is an example of a retrospective cohort study.
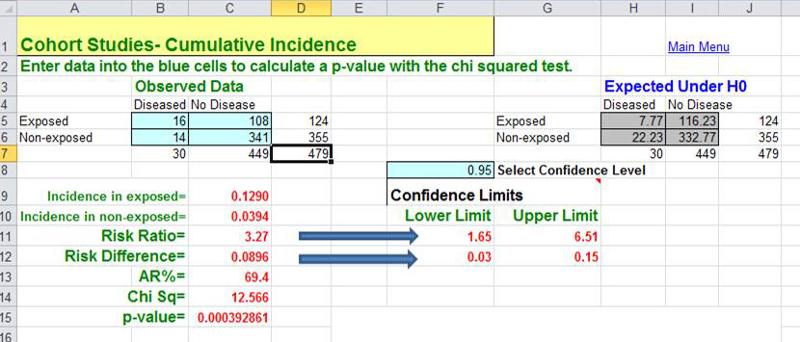
A cohort study may also be ambidirectional , meaning that there are both retrospective and prospective phases of the study. Ambidirectional studies are much less common than purely prospective or retrospective studies, but they are conceptually consistent with and share elements of the advantages and disadvantages of both types of studies. The Air Force Health Study (AFHS) - also known as the Ranch Hand Study - was initiated by the U. S. Air Force in 1979 to assess the possible health effects of military personnel's exposure to Agent Orange and other chemical defoliants sprayed during the Vietnam War. The study was conducted comparing:
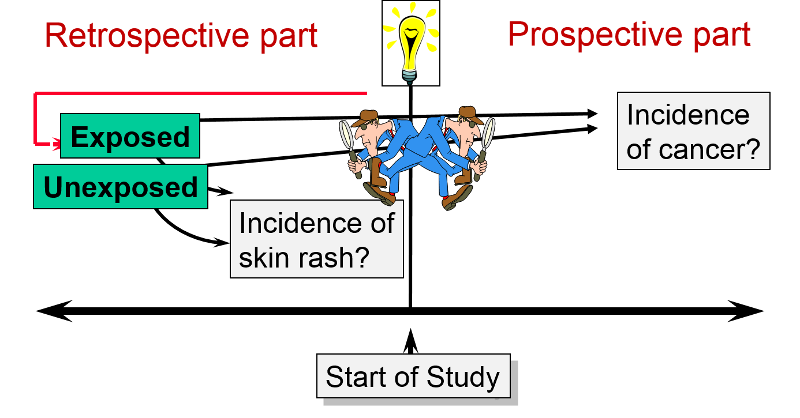
This is an "ambidirectional" study, because it had both a retrospective component and a prospective component. Some of the problems suspected to be caused by Agent Orange would have occurred shortly after exposure (e.g., skin rashes). These were addressed by looking at the cohort retrospectively to see if the exposed pilots had had more problems than the controls. Other problems (e.g., infertility & cancer) might not surface until some time after the exposure. Therefore, the cohort was followed prospectively to see if they had a greater incidence of these problems. The reports that emerged from the study suggested links between Agent Orange exposure and nine distinct diseases: chloracne, Hodgkin's disease, multiple myeloma, non- Hodgkin's lymphoma, porphyria cutanea tarda, respiratory cancers (lung, bronchus, larynx and trachea), soft-tissue sarcoma, acute and subacute peripheral neuropathy, and prostate cancer.
A closed cohort is one with fixed membership. Once the cohort is defined by enrolling subjects and follow up begins, no one can be added. The number of subjects may decline because of death or loss to follow up, but no additional subjects are added. As a result, closed cohorts always get smaller over time. Citizens of Japan who were exposed to radiation when atomic bombs were dropped on Hiroshima and Nagasaki during the second World War, would be considered members of a fixed or closed cohort that was defined by an event. Ashengrau and Seage would classify the bombing victims as a "fixed cohort" and make a distinction between a fixed cohort and a closed cohort. They define a closed cohort as similar to a fixed cohort except that a closed cohort is one that has no losses to follow up, for example, a cohort of people who attended a luncheon that resulted in an outbreak of Salmonellosis.
In contrast, an open cohort is dynamic, meaning that members can leave or be added over time. Rothman gives the example of a state cancer registry. Subjects are continually added when they are diagnosed with cancer, so new subjects are continually added. Subjects can also leave the cohort by moving to a new state or dying. Another example of an open or dynamic cohort would be students at Boston University.
These descriptions should sound familiar, because they essentially parallel the descriptions of fixed and dynamic populations from the Measures of Disease Frequency module. The great majority of cohort studies are conducted in closed (or fixed) cohorts, because it is more difficult to establish eligibility and track people in an open cohort, since they can enter and leave at any time. This problem becomes greater as the size of the cohort gets larger and/or the study continues for a longer period of time. Note that the retrospective cohort study of Giardia in Milton was an open cohort (members of the golf club), but the population was relatively small and time period very short.
For common risk factors, (e.g., smoking, obesity) investigators may enroll a general population cohort, e.g.,
Once a general population cohort is enrolled, investigators will ascertain their baseline exposures to a large number of exposures of interest and possible confounding factors that they may need to adjust for in the analyses.
For uncommon risks, investigators use special exposure cohorts, e.g.,
The ideal comparison group in a cohort study would be a group that was exactly the same as the exposed group, except that they would be unexposed. This is referred to as the "counterfactual ideal," because it is impossible for the same person to be both exposed and unexposed at the same time. Consequently, the best one can do is to select a comparison group that differs with respect to the exposure of interest but is a similar as possible with respect to other factors that might influence the outcome. There are two key things that are essential in selecting the comparison group in a cohort study:
There are three general types of comparison groups for cohort studies.
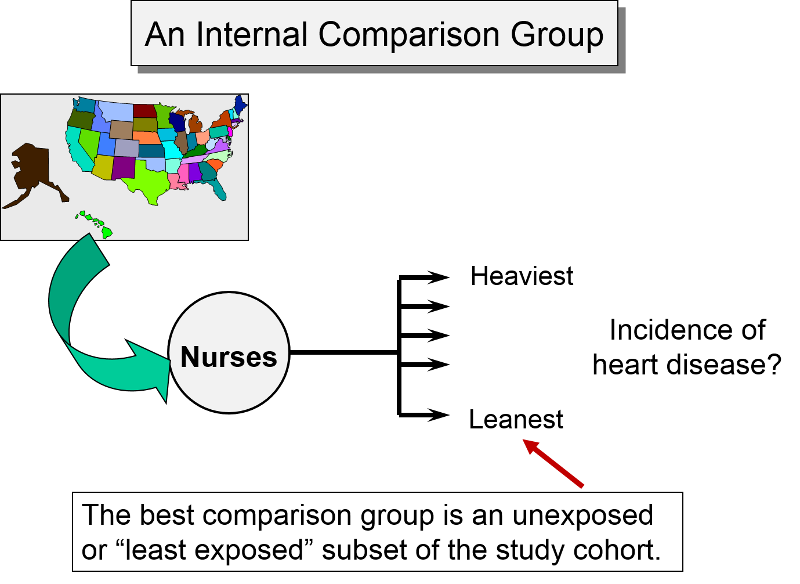
Internal Comparison Group
An internal comparison group consists of unexposed members of the same cohort. This is generally the best comparison group, because the subjects are comparable in many respects. For example, as noted earlier, the Nurses' Health Study, used the cohort to study a possible association between obesity and myocardial infarction. Subjects from the cohort were categorized into one of five levels of body mass index, and the group with the lowest BMI was used as an internal comparison group or reference group, against which the other categories were compared.
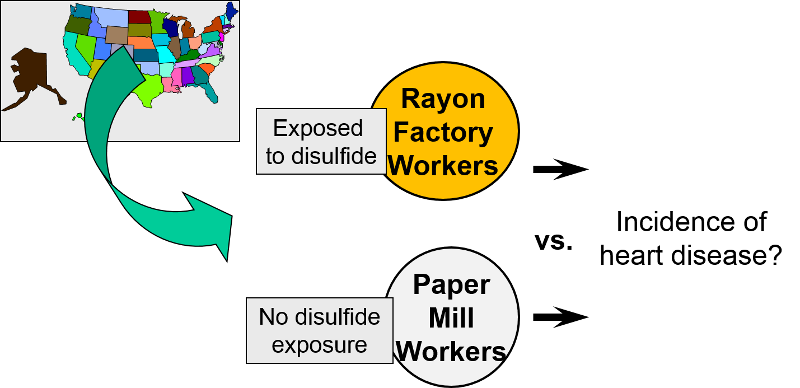
External Comparison Group
When it isn't possible to take a well-defined cohort and divide it into exposure groups, sometimes one can identify an external comparison cohort. This type of comparison group was used when researchers wanted to look at occupational exposure to disulfide in rayon factory workers. Because virtually all workers in the plant were exposed to disulfide, it was not possible to use an internal comparison group from the same plant. Instead, they selected a group of people employed in a paper mill as an external comparison cohort. Both groups had similar education, age, socioeconomic status, and gender distribution. However, they may have differed with respect to other important confounding factors.
General Population as a Comparison Group
The third possibility is to use the general population as a comparison group. This is used occasionally in situations where only a small percentage of the population is exposed, e.g., with an unusual occupational exposure. However, the general population may differ from the exposed work force in many ways, including overal health.
One study looked at mortality rates of workers in the rubber industry and compared them to the general population of the US. There are several problems with this. 1) Some of the general population will have had the exposure (same occupation); 2) the general population includes people who are unable to work because of illness or disability. Employed workers tend to be healthier than the general population. This is a well-documented phenomenon know as the "healthy worker effect." Rates of disease and death tend to be higher in the general population than in the employed work force because the general population includes many people who are too sick or disabled to work. As a result, even if the exposure was, in fact, associated with higher mortality, the magnitude of association would be underestimated because of the inherently higher mortality rate in the general population (which includes both employed and unemployed workers). Although this is not a problem with all diseases, the general population generally exhibits the greatest departure from the counterfactual ideal, and therefore is less widely used today than in the past. Another notable disadvantage of the general population comparison group is that data on important confounders are almost never available.
The use of the general population as a comparison group is most likely to be seen today in descriptive epidemiology, particularly when there are many categories of exposure with only a small number of outcomes per category. For example, the Massachusetts Cancer Registry reports the cancer rate in each of 351 cities and towns using the overall rate in the general population as a comparison. Similarly, descriptions of occupational mortality based on death certificate data may have hundreds of different occupations and also use the general population as a comparison group.
Studies of this type sometimes use a standardized mortality ratio (SMR) as the measure of association. Data from the general population provide overall rates of mortality in the population. These rates are then used to predict how many deaths would be expected in the cohort under study. The SMR is the ratio of observed deaths in the cohort to the number of deaths expected. The SMR is interpreted much like a risk ratio. For example, an SMR=1.2 indicates 1.2 times the risk in the general population or a 20% increase in risk. (Note that sometimes the SMR is multiplied x 100; if so, SMR=120 would also indicate a 20% increase in risk. If population rates are available by age, gender, and race, then SMRs can be adjusted or "standardized" to control for confounding by these factors. This is a method sometimes referred to as "indirect standardization."
A similar analytical approach is used to compute standardized incidence ratios (SIR). For example, the Massachusetts Cancer Registry uses the general population of Massachusetts as a comparison group in order to examine whether the incidence of specific cancers differs in individual communities compared to the entire state's incidence. In this setting the measure of association that is used is a standardized incidence ratio (SIR). SIRs can also be interpreted much like a risk ratio, although they are typically multiplied by 100, so that SIR=120 would indicate an incidence that was 20% greater than that in the overall population.
Link to the Massachusetts Cancer Registry
Link to BUSPH learning module on Standardized Rates and page on Standardized Incidence Ratios
Selection bias from enrollment procedures rarely occurs in cohort studies, because the outcomes have not yet occurred at the time when subjects are enrolled, so a potential participant's eventual outcome status is unknown and therefore can not influence . However, selection bias can occur in a prospective cohort study as a result of differences in retention during the follow-up period after enrollment. When the observation period spans many years (in either retrospective or prospective cohort studies) it can be difficult to track subjects for the entire study. Subjects may disappear as a result of death, relocation, or (in prospective studies) loss of interest in the study. Studies with follow up rates of less than 60% will generally be seen as having limited validity, but even losses of 20% can introduce bias if the reasons for loss are related to both exposure status and outcome status.
Losses to follow-up can introduce bias (a deviation of the observed value of the measure of association from the value that would have been observed in the absence of bias) if there are differences in likelihood of loss to follow-up that are related to exposure status and outcome. In general, large prospective cohort studies are doing well if they can maintain follow-up of 80-90% of their sample for long periods.
Example:
As an illustration of how bias can be introduced with loss to follow-up, consider the following example. Suppose investigators were prospectively studying the association between use of oral contraceptives and development of thromboembolism (TE), i.e., blood clots in veins of the lower extremities or pelvis that can break loose and become lodged in the branches of the pulmonary artery]. Suppose, 20/10,000 OC users developed thromboembolism, but only 10/10,000 controls did, i.e., OC users really had a 2-fold greater risk. If roughly 4,000 subjects were lost to follow-up in each group, and if 12 of the 20 subjects who developed thromboembolism in the OC group became lost to follow-up, but only 2 subjects with thromboembolism were lost in the control group, the differential loss to follow-up would make it appear that rates of thromboembolism were similar, and the estimate of association (risk ratio) would be biased.The bias that can result from this differential follow up is a type of selection bias.
The enrollment of subjects in a prospective cohort study like this would not introduce selection bias, because the outcome has not yet occurred. However, retention of subjects may be differentially related to exposure and outcome, and this has a similar effect that can bias the results. In the hypothetical cohort study below investigators compared the incidence of thromboembolism (TE) in 10,000 women on oral contraceptives (OC) and 10,000 women not taking OC. TE occurred in 20 subjects taking OC and in 10 subjects not taking OC, so the true risk ratio was (20/10,000) / (10/10,000) = 2.
|
Unbiased Results |
Thromboembolism |
Non-diseased |
Totaal |
|---|---|---|---|
|
Oral Contraceptives |
20 |
9980 |
10,000 |
|
Unexposed |
10 |
9990 |
10,000 |
This unbiased data would give a risk ratio as follows:
However, suppose there were substantial loses to follow-up in both groups, and a greater tendency to loose subjects taking oral contraceptives who developed thromboembolism. In other words, there was differential loss to follow up with loss of 12 diseased subjects in the group taking oral contraceptives, but loss of only 2 subjects with thromboembolism in the unexposed group. This might result in a contingency table like the one shown below.
|
Biased Results |
Thromboembolism |
Non-diseased |
Totaal |
|---|---|---|---|
|
Oral Contraceptives |
8 |
5980 |
5988 |
|
Unexposed |
8 |
5984 |
5992 |
This biased data would give a risk ratio as follows:
So, in this scenario both exposure groups lost about 40% of their subjects during the follow up period, but there was a greater loss of diseased subjects in the exposed group than in the unexposed group, and it was this differential loss to followup that biased the results.
A study with substantial loss to follow does not have to produce a biased estimate of an association, but it certaintly raises concerns about the accuracy of the estimate. If the losses among the groups being compared are non-differntial, then the estimate will not be biased by the losses. Since there is no way of predictiing the effects of loss to follow up, researchers do their best to reduce it by maintaining contact with participants at regular intervals, collecting contact information from friends or relatives that would know how to reach a participant should s/he move, using the National Death Index and other databases to track the vital status of participants who do not respond to attempts at contact, as well as other strategies. Most of these strategies are only applicable to prospective cohort studies, because all the follow-up time has already occurred in a retrospective cohort study before the investigators get involved. Carefully done prospective cohort studies will go to extraordinary lengths to maintain high follow-up rates.
|
|
Unlike the bias that can occur from differential loss to follow up, selection bias at enrollment rarely occurs in cohort studies, because the outcomes have not yet occurred at the time when subjects are enrolled, so a potential participant's eventual outcome status is unknown and therefore can not influence their participation. This is even more unlikely in prospective than retrospective cohort studies, although even in the latter the cohort is almost always created based on information that was recorded prior to the development of the outcome.
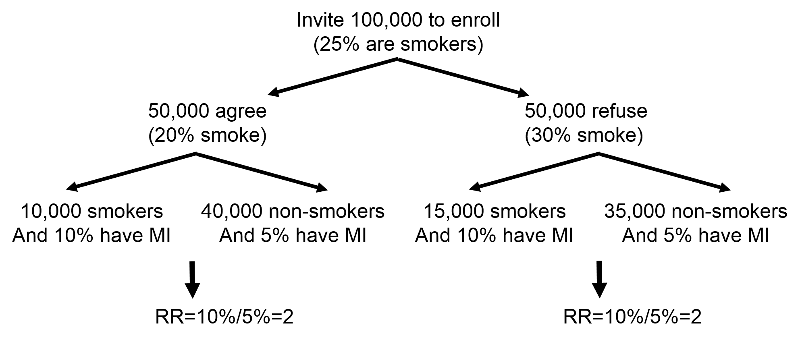
Ordinarily, some of the individuals invited to be subjects in a prospective cohort study refuse to participate. This can produce bias in retrospective cohort studies and case-control studies, because exposure status and outcomes have already occurred at the time of enrollment. However, non-participations will not bias a prospective cohort study in which the outcomes of interest have not yet occurred. To illustrate suppose 100,000 subjects are invited to be in a study on the association between smoking and risk of heart attack, but 50,000 refuse to participate. Only 20% of those who agree are smokers versus 30% in those who refuse. The frequency of smoking in the study subjects is not representative of its prevalence in the population, but you can still compare smokers and non-smokers with respect to the incidence of myocardial infarction. In a situation like this, substantial non-participation could detract from the generalizability of the findings. For example, if there were a high rate of non-participation among African-Americans, it might not be appropriate to extrapolate the findings to this subset of the population.
A similar type of bias can occur in retrospective cohort studies if subjects in one of the exposure groups are more or less likely to be selected if they had the outcome of interest.
Example:
Consider a hypothetical investigation of an occupational exposure (e.g., an organic solvent) that occurred 15-20 years ago in factory. Over the years there were suspicions that working eith the solvent led to adverse health events, but no definitive data existed. Eventually, a retrospective cohort study was conducted using the employee health records. If all records had been retained the results might have looked like those shown in the first contingency table below.
|
Unbiased Results |
Diseased |
Non-diseased |
Totaal |
|---|---|---|---|
|
Solvent exposure |
100 |
900 |
1000 |
|
Unexposed |
50 |
950 |
1000 |
This unbiased data would give a risk ratio as follows:
However, suppose that many of the old records had been lost or discarded, but,given the suspicions about the effects of the solvent, the records of employees who had worked with the solvents and subequently had health problems were more likely to be retained. Consequently, record retention was 99% among workers who were exposed and developed health problems, but recorded retention was only 80% for all other workers. This scenario would result in data shown in the next contingency table.
|
Biased Results |
Diseased |
Non-diseased |
Totaal |
|---|---|---|---|
|
Solvent exposure |
99 |
720 |
819 |
|
Unexposed |
40 |
760 |
800 |
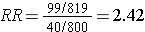
Differential loss of records results in selection bias and an overestimate of the association in this case, although depending on the scenario, this type of selection bias could also result in an underestimate of an associaton.
|
|
Clarity of Temporal Sequence (Did the exposure precede the outcome?): Cohort studies more clearly indicate the temporal sequence between exposure and outcome, because in a cohort study, subjects are known to be disease-free at the beginning of the observation period when their exposure status is established. In case-control studies, one begins with diseased and non-diseased people and then acertains their prior exposures. This is a reasonable approach to establishing past exposures, but subjects may have difficulty remembering past exposures, and their recollection may be biased by having the outcome (recall bias).
Allow Calculation of Incidence: Cohort studies allow you to calculate the incidence of disease in exposure groups, so you can calculate:
Facilitate Study of Rare Exposures: While a cohort design can be used to investigate common exposures (e.g., risk factors for cardiovascular disease and cancer in the Nurses' Health Study), they are particularly useful for evaluating the effects of rare or unusual exposures, because the investigators can make it a point to identify an adequate number of subjects who have an unusual exposure, e.g.,
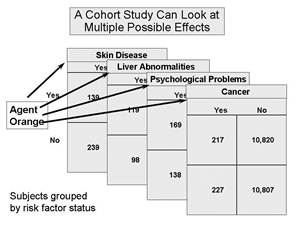
Allow Examination of Multiple Effects of a Single Exposure
Avoid Selection Bias at Enrollment: Cohort studies, especially prospective cohort studies, reduce the possibility that the results will be biased by selecting subjects for the comparison group who may be more or less likely to have the outcome of interest, because in a cohort study the outcome is not known at baseline when exposure status is established. Nevertheless, selection bias can occur in retrospective cohort studies (since the outcomes have already occurred at the time of selection), and it can occur in prospective cohort studies as a result of differential loss to follow up.
The "Air Force Health Study" on agent orange illustrates these advantages.
Link to a video on Agent Orange from the the New York Times

Pitfalls:
The video below (21 min.) will demonstrate how to analyze a large cohort study for which data has been stored in an Excel spreadsheet.
Moderate Exercise or Just Take a Statin?
This exercise is based on the following two journal articles that focus on strategies to prevent cardiovascular disease. Links to the full articles are provided below.
Compare and contrast the potential utility of each of these interventions as strategies to reduce cardiovascular disease in a population. In your discussion you should address issues of feasibility, cost, adherence, and risk versus benefit. Would you favor one intervention over the other? Why?